When a surgeon removes lymph nodes to stop cancer spreading, they solve one problem and create another. The body can’t replace what’s gone. For hundreds of thousands of patients worldwide, what follows is decades of swelling limbs, chronic infections, and the constant burden of compression garments.
But what if we could simply build the missing piece?
Kosuke Kusamori at Tokyo University of Science had a radical idea: skip the complexity. Instead of trying to coax the body into regenerating lymph nodes through genetic engineering or stem cell transplantation, approaches that have largely failed, why not just construct one from scratch using simple biology? This month, his team reported something that seemed impossible. They’ve created a replacement lymph node made from human cells, transplanted it into mice missing their own, and watched as it restored normal limb function within weeks.
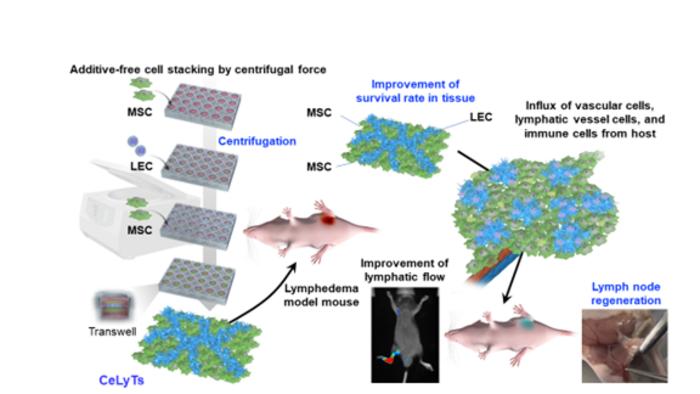
The technique is almost deceptively straightforward. Take two types of cells: lymphatic endothelial cells, which line lymph vessels, and mesenchymal stem cells, which provide structural support. Layer them in the precise arrangement nature uses, using nothing more exotic than centrifugal force from a standard laboratory centrifuge. Culture the resulting three-layered structure for a few days until the cells organize into a network. Then transplant. What emerges in the body is a functional replacement that filters fluid, mounts immune responses, and behaves like an actual lymph node.
The problem being solved is ancient and familiar. Every year, tens of thousands of cancer patients undergo lymphadenectomy (surgical removal of lymph nodes to prevent malignant cells from spreading). The procedure saves lives. But lymph nodes don’t regenerate. Their absence breaks the body’s drainage system. Fluid accumulates in the affected limb, triggering chronic inflammation, thickened skin, fatty tissue accumulation, and recurrent infections. Secondary lymphedema affects an estimated one in five breast cancer survivors. Existing treatments, including compression garments, manual drainage, and occasionally surgical rerouting, manage symptoms but never cure the underlying problem.
Researchers have tried everything to regenerate lymph nodes. Gene therapy approaches introducing growth factors showed promise on paper but failed in practice. Stem cell transplantation seemed logical but produced no functional improvement. Even tissue engineering, the theoretical solution, mostly delivered lymph vessels without actually reconstructing nodes. The field had stalled at a fundamental impasse: how do you make a lymph node rather than just lymphatic vessels?
Kusamori’s insight was to recognize that complexity might be the enemy. Instead of building complexity into the engineering process, his team used centrifugal force to organize cells into the precise geometry nature requires. The three-layer architecture matters: lymphatic cells on the inside forming the filtering network, stem cells on the outside providing immune support and preventing rejection. Culture it briefly to allow internal organization. That’s the entire process.
When tested in mice with surgically removed lymph nodes, the engineered tissues (called CeLyTs for centrifuge-based bioengineered lymphatic tissues) worked immediately. Within days of transplantation, new blood vessels grew around the graft. Within ten days, the structure had integrated with the body’s own cells and begun functioning as a proper lymph node. The mice’s limbs, which had swollen dramatically after lymph node removal, returned to normal thickness. Their immune response improved. Fatty tissue that had accumulated in the skin began to resolve.
The comparison to standard care is striking. Compression therapy, the gold standard for decades, merely slows swelling temporarily. It requires lifelong application of tight wraps or garments—uncomfortable, limiting, but necessary. Kusamori’s team found that CeLyTs suppressed lymphedema far more effectively than compression, and they did something compression cannot: they reversed the underlying damage. Even in a severe chronic model where mice had multiple lymph node regions removed, CeLyTs controlled swelling and restored immune function better than other tissue engineering approaches.
The engineered structures themselves prove remarkably sophisticated. When Kusamori’s team examined them under the microscope weeks after transplantation, they found complex architecture: follicle-like regions where immune cells congregated, sinus-like spaces for fluid processing, zones of tissue organization. The structures were composed of both transplanted and host-derived cells; they had essentially instructed the body to participate in their own maturation. The immune cells present—T cells, B cells, macrophages, dendritic cells—weren’t just passing through. They organized into the patterns found in natural lymph nodes and mounted appropriate immune responses.
None of this required immunosuppressive drugs. The mesenchymal stem cells present in the tissue naturally suppress rejection, allowing the human-derived structures to survive and function in immunodeficient mice. In actual clinical translation, that advantage could be transformative. Patients with organ transplants accept a lifetime of medication to prevent rejection, with all its associated complications. An engineered lymph node that engineered its own tolerance would eliminate that burden.
The mathematics of treatment cost alone suggest clinical promise. A single transplantation could provide sustainable benefit for years, possibly decades. No repeated hospital visits. No accumulated years of compression garment purchases. One procedure to address a condition that currently means permanent management of symptoms. Even the tissue manufacturing is elegantly simple. The centrifugal cell stacking process requires no specialized additives, no complex scaffolding, no proprietary components. It’s scalable.
The work reveals something important about engineering biology: sometimes the breakthrough isn’t in making the engineering process more complicated. It’s in stepping back and asking whether we’ve overcomplicated what nature already understands how to do. Kusamori and his colleagues essentially asked the body’s own design principles what the right solution looked like, then got out of the way.
Secondary lymphedema may finally have met its match. For patients who have already survived cancer, that could mean something profound: not just managing a condition, but erasing it. The engineered lymph node wasn’t supposed to work. The body wasn’t supposed to let foreign cells establish themselves without constant immunosuppression. Simple centrifugal force wasn’t supposed to organize cells into complex functional structures. Yet here it is, a replacement organ that grows from three layers of cells and the force that’s been spinning samples in research labs for a century.
Study link: https://www.nature.com/articles/s41467-025-65121-3
If our reporting has informed or inspired you, please consider making a donation. Every contribution, no matter the size, empowers us to continue delivering accurate, engaging, and trustworthy science and medical news. Independent journalism requires time, effort, and resources—your support ensures we can keep uncovering the stories that matter most to you.
Join us in making knowledge accessible and impactful. Thank you for standing with us!